Quality risk management from the beginning of the planning phase
The requirements outlined by the relevant European GMP guidelines and EHEDG guidelines must be met for systems that process food, food supplements or basic and active ingredients for the food, cosmetics or pharmaceutical industries. The focus is always on ensuring product quality. As part of quality risk management, the risks to product quality are identified and minimized during the planning, construction and operation of the plant.
As an active member of the European Hygienic Engineering & Design Group EHEDG, Glatt is not only involved in the development of new guidelines, but also consistently implements them in clever hygiene concepts. The design and material of our systems and the surface quality of components that come into contact with the product are therefore just as well thought out as the cleaning processes themselves.
Glatt integrates the quality risk management approach into its system planning and construction:
- Focus on GMP or hygiene concept at the beginning of the plant planning, this includes all requirements that are relevant for ensuring product quality
- Hygienic design of the system in accordance with the required product quality: materials, surfaces, cleanability
- Hygienic components, measuring devices, connections, pipework and cable routing
- Cleaning concept development according to product characteristics and operating regime
- Design of the system environment as a hygienic or cleanroom area
- Confirming the requirements have been met during FAT and commissioning, and even qualification of the system if necessary
- Development of a maintenance and servicing strategy
With this approach, we will find suitable, cost-effective and well-documented solutions for you to fulfil user requirements and comply with the applicable regulations. You receive a high-quality system with appropriate documentation, a reliable basis for operation and maintenance and thus the best conditions for high and consistent product quality.
Interaction of cleaning concept and hygiene components to ensure product quality and avoid cross-contamination
The design parameters of the fluidized bed or spouted bed production system depend largely on the quality requirements of the end products and their use.
A well thought-out system concept can significantly influence capital expenditure. To do this, it is first necessary to know or determine the exact requirements for the end products. Glatt has developed a classification system that focuses on the cleaning requirements rather than the conventional categorization of the product. For example, with an intelligent design for easy cleaning and a well-planned room and zone concept for wet cleaning of the system and dry storage of filter elements, product quality in production can be ensured in the same way as in a classic hygiene-compliant design – but the costs are lower.
As a long-standing plant manufacturer, we are aware of the importance of factors for cleaning success and supply systems that also fulfil special hygiene and cleaning requirements. Based on our experience and customer knowledge of the properties of raw materials and products with regard to cleanability and the risks of cross-contamination, we develop customized solutions. The cleaning procedure is optimized in terms of time, temperature, mechanics and concentration of the cleaning media (alkalis, acids or industrial cleaners).
Accessible filter chamber
Manual dry or wet cleaning may be sufficient for single-product systems. Frequent product changes or the requirement to avoid cross-contamination increase the cleaning effort. Semi-automated cleaning (Washing in Place, WIP) or fully automated cleaning (Cleaning in Place, CIP) is advisable in this case.
We offer customized cleaning systems. We can draw on the Glatt Group’s many years of experience, ranging from Washing in Place (WIP) systems to validatable Cleaning in Place (CIP) systems. We attach great importance to optimizing the cleanability of all systems. This includes easy access to all parts of the system as well as tried-and-tested cleaning openings and water drains and the installation of cleaning nozzles.
Many design details need to be considered in order to realize partially or fully automated cleaning of the system:
- Hygienic design to ensure complete drainability
- High surface quality of the parts in contact with the product
- Spray nozzles are easy to dismantle
- Correct arrangement of the cleaning nozzles
- Special sensors to monitor the success of the cleaning process
Our WIP/CIP cleaning systems supply your process systems with all the necessary cleaning media with unlimited flexibility
Glatt supplies cleaning systems to match the cleaning concept. Everything is possible, from standard designs to customized solutions. For batch systems in particular, we offer ready-made solutions for individual cleaning modules through to central cleaning stations for multiple systems. You can choose between semi-automatic WIP cleaning or fully automatic CIP cleaning, which is validated on the basis of defined success criteria. Semi-automatic WIP cleaning is the standard for continuously operated systems.
Glatt’s cleaning systems can be flexibly customized to your requirements. For example, continuous or circulation cleaning is available. The dosing of one or more cleaning agents can be carried out with standardized cleaning dosing stations. We offer the option of expanding the basic version with customized additional modules to meet your requirements.
Glatt cleaning systems are also flexible when it comes to the control system: whether stand-alone or integrated control, whether manual, semi-automatic or fully automatic cleaning – you get the control system that suits your requirements.
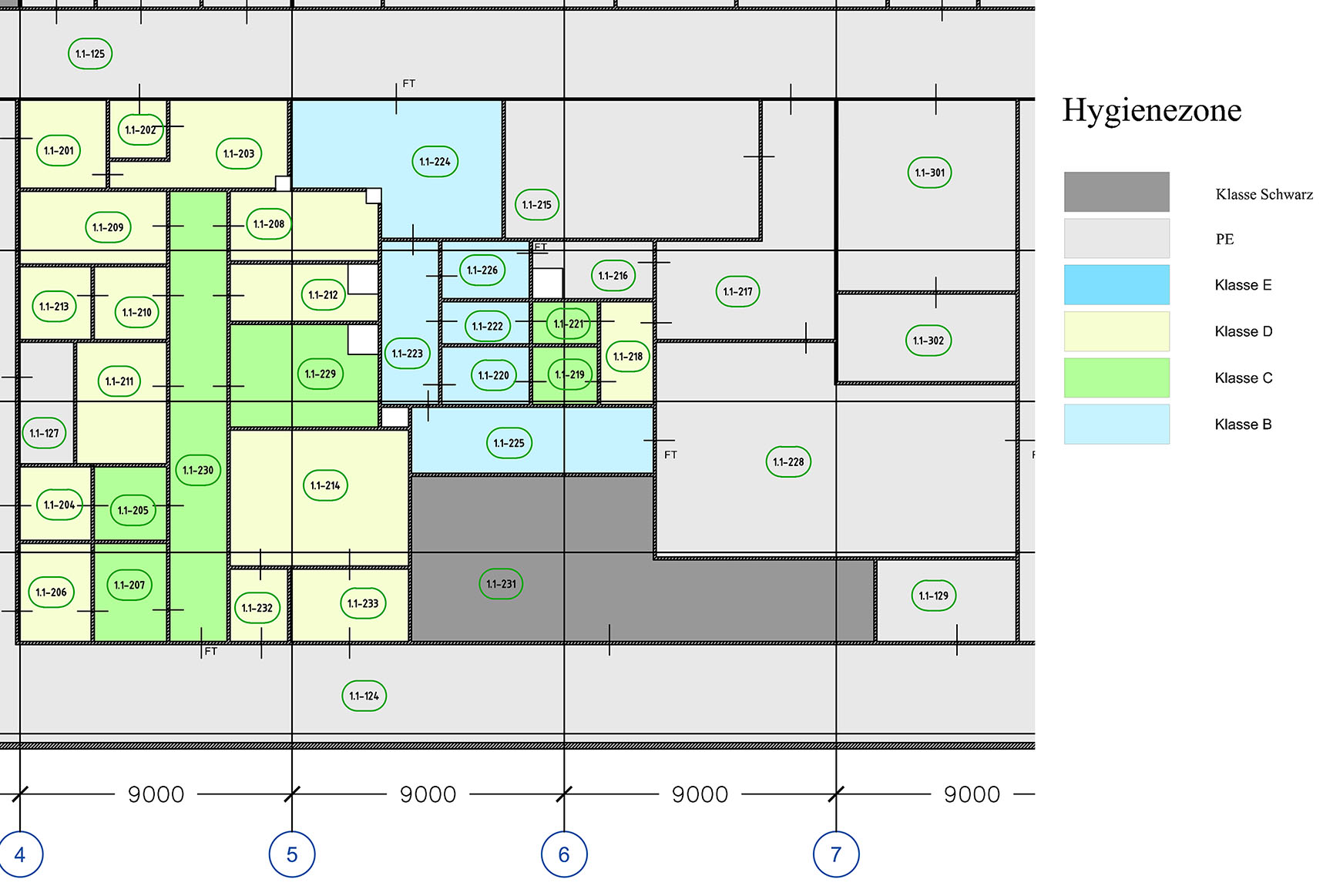
Zone concepts according to GMP offer a high level of safety to protect products from contamination and cross-contamination.
Stainless steel stage, Glatt hygienic design
Panel feed-through, Glatt hygienic design
Further information on this topic and related topics can also be found in the following publications:
Published article: ‘Everything according to the rules – Hygiene design for fluid bed and spouted bed systems’ PDF, German
Published article: ‘Product safety for granulation and coating – Hygienic design for fluid bed and spouted bed systems’ PDF, German
Published article: ‘Better safe than sorry – Design safety for fluid bed systems’ PDF, German
Published article: ‘Cleanly separated – filters in fluid bed systems, an overview’ PDF, German
Published article: ‘Fluid bed systems: continuous, batch or a hybrid?’ PDF, German